|
Therefore, the lone pair exhibits a polar character. A lone pair is involved in changing the angle of the molecule.The lone pair is involved in determining the geometry and shape of the molecule, and it is an essential part of the electron domain. Valance electrons in atom = Lone pair + Bond pair To calculate the number of valance electrons in an atom, we add lone pair in the bonding electron pair. We can identify the lone pair by using the Lewis structure. It is also called a non-bonded electron pair or unshared pair of electrons. 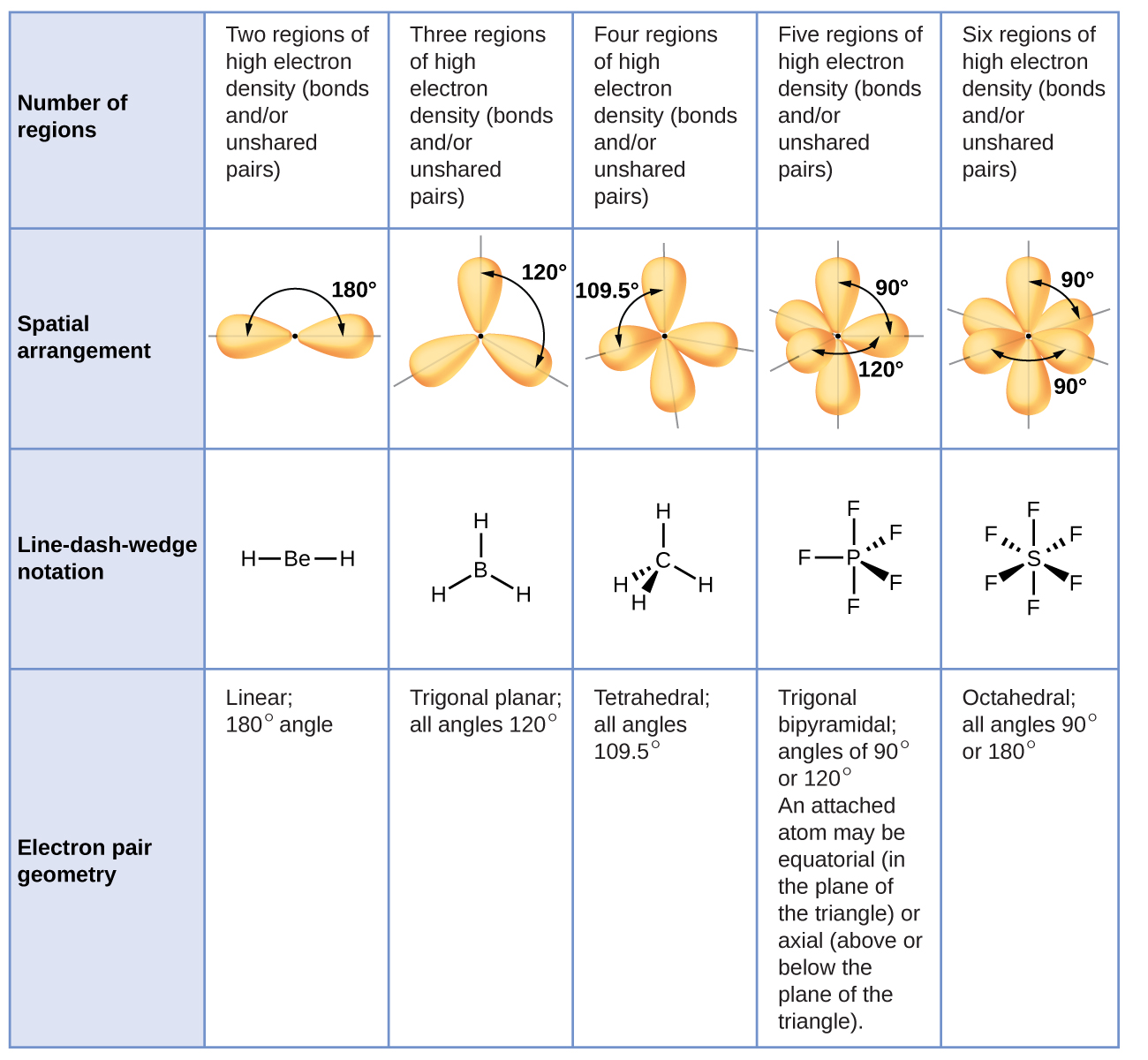
It is connected to an atom by using a coordinate covalent bond. A pair of electrons that is not reacted or shared by an atom is called lone pair. lone pair and bond pair What is Lone pair?Ī lone pair is associated with the negative charge. The diagram represents the electron domains i.e. So, the repulsion of electrons is not only the factor that describes molecular geometries. electrons and positively charged nuclei attract towards eachother.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
